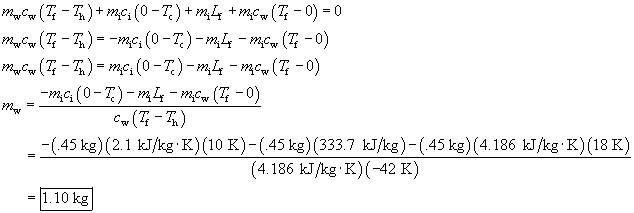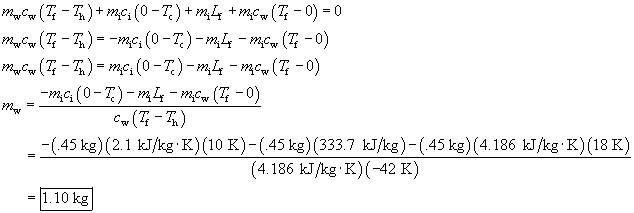
1. Identify all the variables. Use subscripts that clearly distinguish these quantities (e.g., cw for the specific heat of water, cc for the specific heat of the container, etc.).
2. List all the heat exchange (energy transfer) processes that occur; note whether or not there is a temperature change and whether the sign of the change is positive or negative. If a sample changes temperature (gets warmer or cooler), the energy term is +mc(Tf - Ti), where c is the appropriate specific heat.If the sample gets cooler, the temperature difference will be negative, since Tf < Ti.
Be especially careful to identify the correct sign for changes of state, involving “latent heat.” If a solid sample melts, the energy term is +mLf. If a sample in the liquid state solidifies (e.g. water freezes), the energy term is -mLf. If a liquid vaporizes (e.e. water boils), the energy term is +mLv. If a vapor condenses to liquid form, the energy term is -mLv.
3. Write the conservation of energy equation, in symbolic form (no numbers!), making sure that there is a term for each of the processes identified in step 2. Set the sum of all the energy exchange terms equal to zero. In processes for which a temperature change occurs, make sure that the change is expressed as (final temperature - initial temperature).
4. Where appropriate, expand parentheses. E.g., write mwcw(Tf - Th) as mwcwTf - mwcwTh.
5. Solve algebraically for the unknown quantity.
6. Substitute numerical values, including units, and calculate.
7. Make sure that the answer is expressed in the correct units, with the required number of significant figures, and is physically reasonable (e.g., the temperature of liquid water must be between 0° C and 100° C). You may even want to substitute your answer in the original conservation of energy equation to see if all the terms add to zero (allowing for round-off error).
Example.
An ice block with a mass of 0.45 kg is initially at a temperature of - 10° C. It is placed in warm water, initially at 60° C, in a well insulated container. The final temperature of the mixture is 18° C. Calculate the quantity of water required for this process. Assume negligible heat transfer to the container.
Solution.
mi = .45 kg, Tc
= -10° C, Tf = 18° C, Th
=
60° C
| Process |
Energy Term |
Sign |
| Warm water cools from Th = 60° C to Tf = 18° C | mwcw(Tf _ Th) | (_) |
| Ice warms from Tc = -10° C to T = 0° C | mici(0 _ Tc) | (+) |
| Ice melts at T = 0° C | miLf | (+) |
| Melted ice warms from T = 0° C to Tf = 18° C | mwcw(Tf _ 0) | (+) |